Conservation Laws in Physics
Overview
Conservation Laws in Physics explains the fundamental quantities that remain constant during physical processes in isolated systems.
Conservation laws are powerful because they allow prediction of outcomes even when the detailed mechanism is complicated.
They are especially important in:
- mechanics
- collisions
- electric circuits
- waves
- nuclear reactions
- particle interactions
In H2 Physics, conservation laws are widely used to solve problems efficiently.
Core Ideas
- conservation laws place strong restrictions on what can happen in a physical process
- energy can be transferred or transformed but total energy remains constant in an isolated system
- momentum is conserved in an isolated system with no external resultant force
- charge is conserved in electric and nuclear processes
- nucleon number and proton number must balance in H2 nuclear equations
- mass is not separately conserved in modern physics; the deeper law is mass-energy conservation
Why Conservation Laws Matter
A physical process may involve:
- forces changing motion
- energy transfer
- particle transformation
- collisions
- decay
Even when details are complex, certain total quantities remain constant.
This provides strong constraints on what can and cannot happen.
Main Conservation Laws in H2 Physics
1. Conservation of Energy
Energy cannot be created or destroyed.
It can only be transferred or transformed.
Examples:
- gravitational potential energy to kinetic energy
- chemical energy to thermal energy
- electrical energy to light
- mass defect to nuclear energy
General idea:
For the broader treatment of energy forms and transfers, see Energy Forms and Conservation and Work, Energy and Power.
2. Conservation of Linear Momentum
In an isolated system with no external resultant force:
Where momentum is:
Momentum is a vector, so direction matters.
It is used in:
- collisions
- explosions
- recoil
- nuclear decay
For more on system-level collision reasoning, see Momentum Conservation and Collisions.
3. Conservation of Charge
Total electric charge remains constant.
Example:
Before:
- neutral atom = 0
After ionisation:
- positive ion + electron
Total charge remains unchanged.
It is used heavily in:
- electric circuits
- electrostatics
- particle decay
- nuclear equations
4. Conservation of Nucleon Number
In nuclear reactions, the total nucleon number is conserved at H2 level.
Where:
- proton = 1 nucleon
- neutron = 1 nucleon
Example:
Top numbers:
5. Conservation of Proton Number
Total proton number , equivalently total charge, is conserved.
Example:
Bottom numbers:
Conservation of Mass
In classical physics, mass is often treated as conserved.
However, in modern physics:
- mass may convert to energy
- energy contributes to the total mass-energy accounting
So the deeper law is mass-energy conservation:
This matters especially in nuclear physics.
Applications in Mechanics
Collisions
For isolated systems:
Types of Collision
Elastic Collision
- momentum conserved
- kinetic energy conserved
Inelastic Collision
- momentum conserved
- kinetic energy not conserved
Explosions and Recoil
Initial momentum may be zero.
After separation:
- total momentum is still zero
Example:
Gun recoil:
- bullet forward momentum
- gun backward momentum
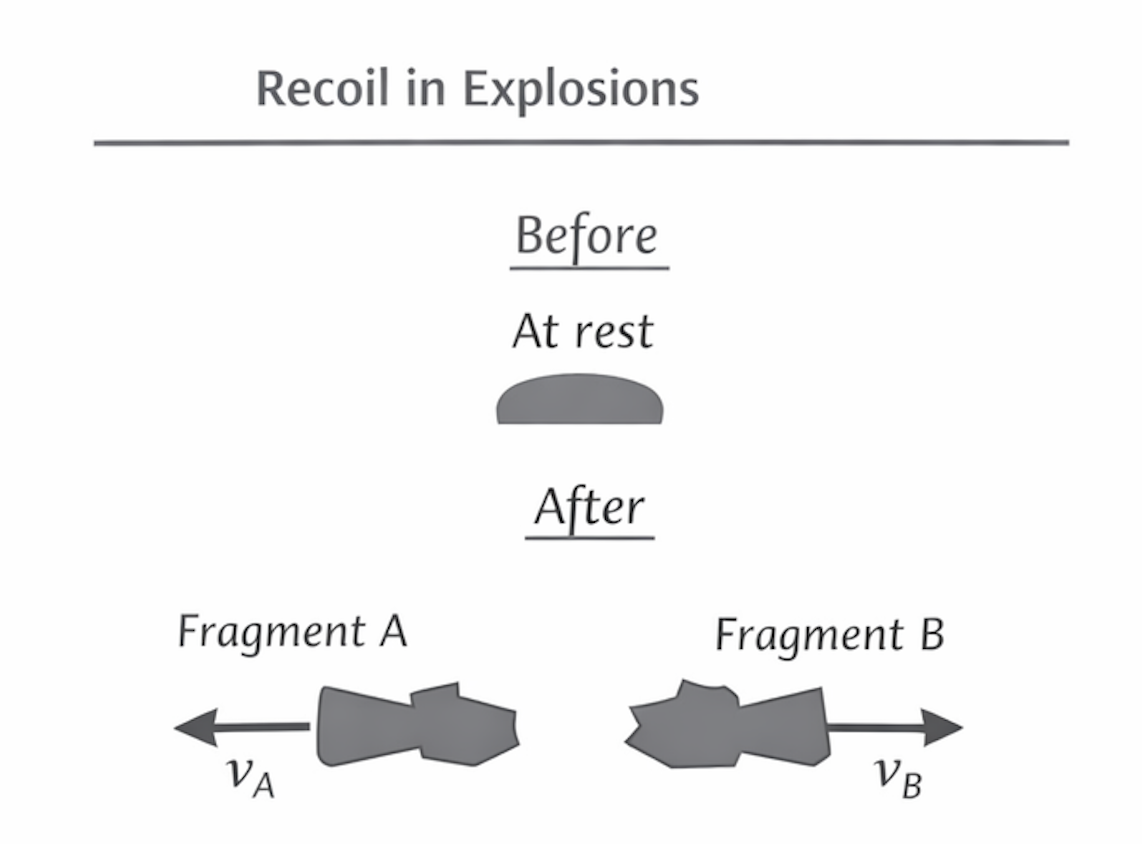
Figure: Recoil after an explosion.
Applications in Nuclear Physics
Conservation laws determine valid nuclear equations.
Example: Alpha Decay
Check:
- nucleon number conserved
- proton number conserved
- energy conserved
- momentum conserved
Example: Beta Decay
The antineutrino helps conserve:
- energy
- momentum
A full lepton-number treatment is beyond the H2 scope of this wiki.
Applications in Circuits
Charge Conservation at a Junction
Current entering a junction equals current leaving it.
Kirchhoff’s first law:
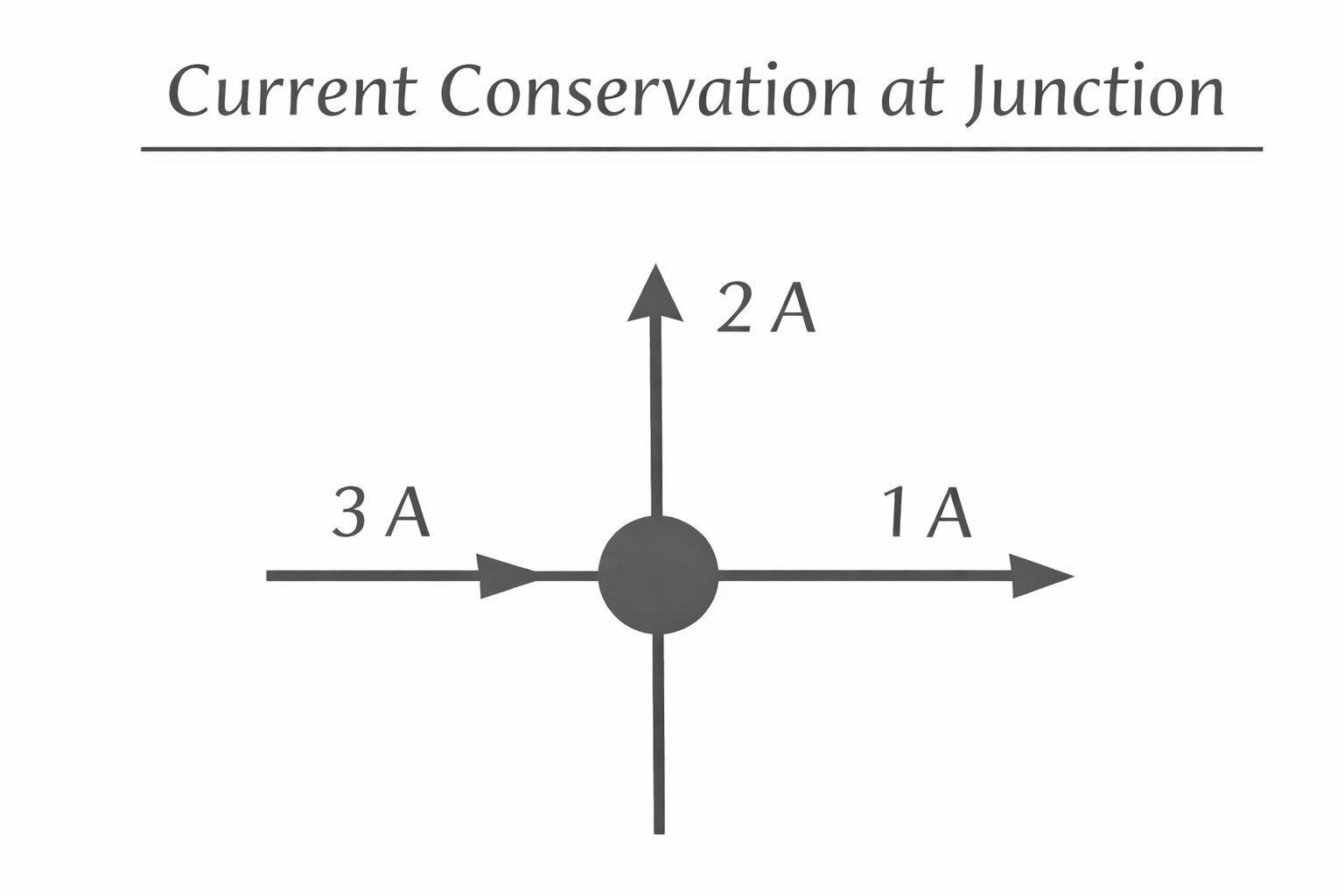
Figure: Current conservation at a junction.
This is used heavily in DC Circuits.
Applications in Waves
Energy Conservation
Wave energy is transferred from a source to a medium or field.
Examples:
- electrical energy to sound
- electrical energy to electromagnetic radiation
How to Use Conservation Laws in Questions
Step 1: Identify the System
Choose the object, collection of objects, or particles involved.
Step 2: Identify Whether It Is Isolated
Ask whether significant external forces or external energy transfers are present.
Step 3: Choose the Relevant Law
Ask which of these is useful:
- momentum
- energy
- charge
- nucleon number
- proton number
Step 4: Write Before = After
Set up equations carefully.
Common Exam Examples
Example 1: Collision
Two carts stick together.
Use:
- momentum conserved
Do not assume kinetic energy is conserved.
Example 2: Nuclear Equation
Use:
- :
- :
Answer:
Example 3: Junction Current
3 A enters a node. One branch carries 1 A.
Remaining branch:
Exam Relevance
This topic is especially useful for:
- nuclear equations
- radioactive decay processes
- fission and fusion reactions
- collisions and recoil
- current-junction reasoning
- energy-transfer problems
Common mistakes include:
- applying momentum conservation to one object instead of a system
- assuming kinetic energy is always conserved
- forgetting direction in momentum
- forgetting charge balance in beta decay
- treating mass as separately conserved in nuclear reactions
Quick Comparison Table
| Quantity | Conserved When |
|---|---|
| Energy | Isolated system when all forms are counted |
| Momentum | No external resultant force on the system |
| Charge | Always observed |
| Nucleon number | Nuclear reactions at H2 level |
| Proton number | Nuclear reactions |
Quick Revision Summary
- conservation laws strongly restrict physical outcomes
- energy can transform but total remains constant
- momentum is conserved in isolated systems
- charge is conserved
- nuclear equations must conserve and
- conservation laws help solve unseen problems efficiently
Links
- Prerequisite: energy forms and conservation
- Prerequisite: dynamics
- Related: work energy and power
- Related: dc circuits
- Related: momentum conservation and collisions
- Related: nuclear physics
- Related: radioactive decay
- Related: particle physics
- Related: nuclear fission
- Related: nuclear fusion