Measurement
Overview
Measurement is the foundation of experimental physics. Every physical law is tested through observations and measured data. Good measurement requires:
- correct units
- sensible precision
- awareness of uncertainty
- clear presentation of data
- fair experimental design
This topic supports all later chapters such as Kinematics, Forces, and Current Electricity Fundamentals.
A strong understanding of measurement helps students avoid common practical and examination mistakes.
Core Ideas
Physical Quantities
A physical quantity is a measurable property that can be expressed by:
- a numerical value
- a unit
Examples:
- length =
- time =
- mass =
Physical quantities are divided into:
- base quantities
- derived quantities
SI Base Quantities and Units
| Base Quantity | SI Unit | Symbol |
|---|---|---|
| length | metre | m |
| mass | kilogram | kg |
| time | second | s |
| electric current | ampere | A |
| thermodynamic temperature | kelvin | K |
| amount of substance | mole | mol |
| luminous intensity | candela | cd |
For H2 Physics, the most frequently used are .
Derived Quantities and Units
Derived quantities are formed from base quantities.
Examples:
| Quantity | Expression | Unit |
|---|---|---|
| velocity | displacement / time | m s |
| acceleration | velocity / time | m s |
| force | mass × acceleration | N |
| energy | force × distance | J |
| pressure | force / area | Pa |
| charge | current × time | C |
Named units:
See Measurement Units and Dimensions.
Dimensions and Homogeneity
Dimensions describe the physical nature of quantities using symbols such as:
- Mass:
- Length:
- Time:
- Current:
Examples:
- velocity:
- acceleration:
- force:
Principle of Homogeneity
All terms in a valid physical equation must have the same dimensions.
Example:
- has dimension
- has dimension
Hence the equation is dimensionally consistent.
Dimensional checks help detect mistakes, but do not prove an equation is physically correct.
Prefixes and Orders of Magnitude
Common SI Prefixes
| Prefix | Symbol | Value |
|---|---|---|
| pico | p | |
| nano | n | |
| micro | ||
| milli | m | |
| centi | c | |
| kilo | k | |
| mega | M | |
| giga | G |
Examples:
Order of Magnitude
Approximate power of ten estimate.
Examples:
- diameter of atom
- human height
- Earth radius
See Measurement Estimation and Experimental Design.
Precision vs Accuracy
Precision
How close repeated readings are to one another.
- small spread of values
- related to random error
Accuracy
How close a reading is to the true or accepted value.
- related to systematic error
Example
Readings of
- high precision
If true value is :
- also high accuracy
A set of readings can be:
- precise but inaccurate
- accurate on average but imprecise
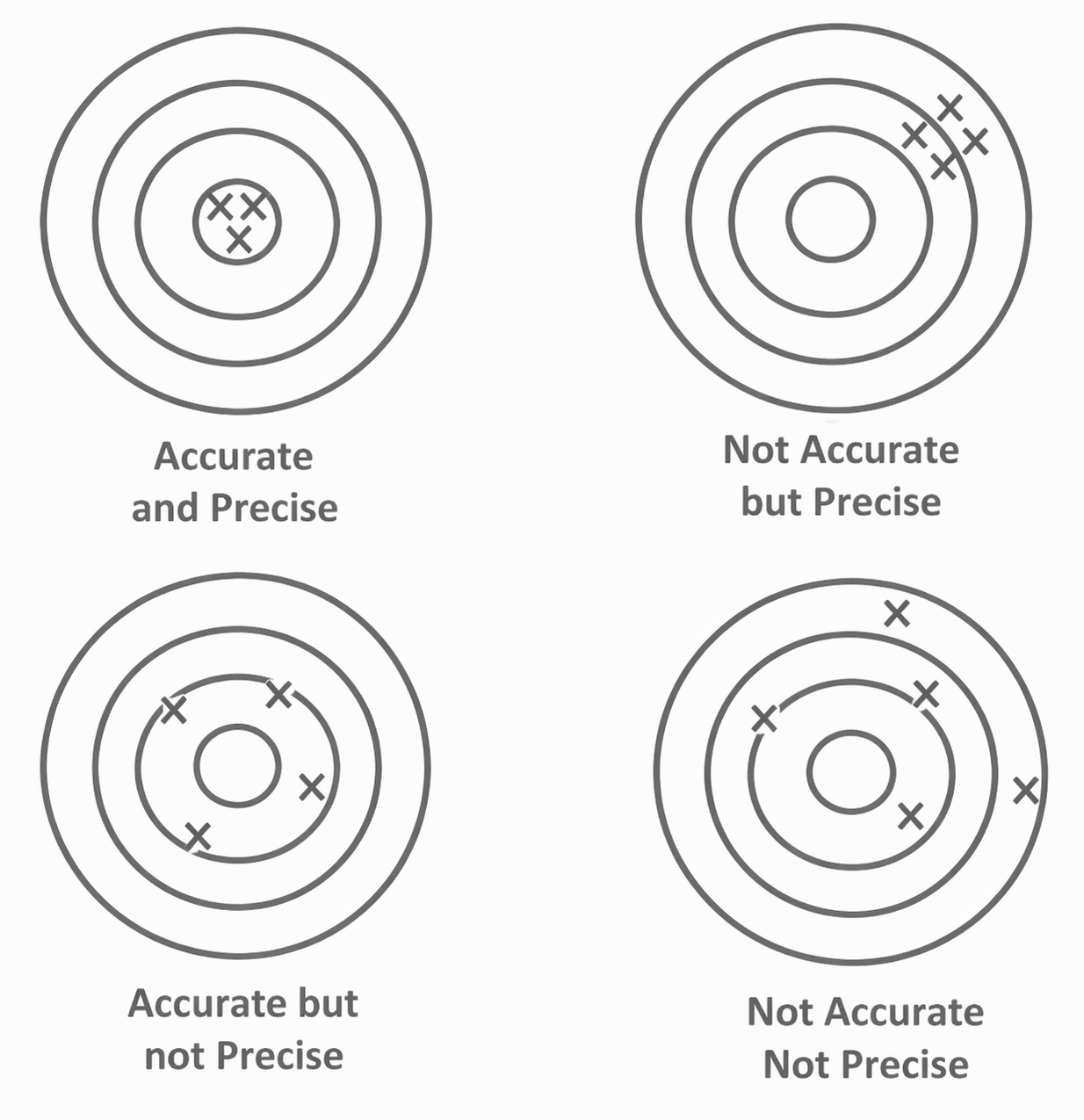
Figure: Accuracy and precision are different. Measurements may cluster tightly without being close to the true value, or scatter around the true value without being precise.
Systematic Errors vs Random Errors
Systematic Errors
Errors that shift readings consistently in one direction.
Examples:
- zero error
- poor calibration
- heat loss in calorimetry
- friction ignored
Effects:
- reduces accuracy
- repeating readings does not remove it
Random Errors
Unpredictable fluctuations between readings.
Examples:
- reaction time
- changing surroundings
- reading scale by eye
Effects:
- reduces precision
- reduced by repeated readings and averaging
See Measurement Uncertainty and Errors.
Uncertainty Overview
Every measured value has uncertainty.
Example:
This means likely range:
Fractional Uncertainty
Percentage Uncertainty
Example:
Percentage uncertainty:
Propagation
When values are combined, uncertainties combine too.
See Uncertainty Propagation Methods.
Data and Graph Overview
Experimental data must be presented clearly.
Tables
Use headings with quantity and unit.
Example:
| 0.0 | 0.00 |
| 1.0 | 2.10 |
| 2.0 | 4.20 |
Graphs
Include:
- title if needed
- labelled axes
- units
- sensible scales
- best-fit line/curve
Useful Interpretation
- gradient often represents physical quantity
- intercept may have meaning
Example:
Velocity-time graph gradient = acceleration.
See Measurement Data Presentation.
Estimation and Experimental Design Overview
Physics students should be able to estimate sensible values and plan fair tests.
Good Experiment Design Includes:
- suitable instrument choice
- control of variables
- repeated measurements
- reduction of uncertainty
- clear method
- safe procedure
Example
To measure pendulum period:
- time 20 oscillations instead of 1
- divide by 20
This reduces reaction-time uncertainty.
See Measurement Estimation and Experimental Design.
Vector Bridge Note
Some older syllabuses introduced vectors within measurement. In this wiki, vectors are developed separately in:
This keeps the Measurement chapter focused on experimental skills and data handling.
Formula Summary
Density
Speed
Fractional Uncertainty
Percentage Uncertainty
Gradient
Common Exam Mistakes
- omitting units
- wrong unit prefixes
- confusing precision with accuracy
- confusing systematic and random errors
- too many significant figures
- graph axes not labelled
- using poor scale
- drawing line through every point instead of best-fit line
- forgetting uncertainty in final answer
Fast Revision Checklist
You should be able to:
- state SI base quantities and units
- convert prefixes
- determine derived units
- check homogeneity
- explain precision vs accuracy
- distinguish systematic vs random errors
- calculate percentage uncertainty
- present tables and graphs properly
- describe methods to reduce uncertainty
Related Links
- Measurement Units and Dimensions
- Measurement Uncertainty and Errors
- Uncertainty Propagation Methods
- Measurement Data Presentation
- Measurement Estimation and Experimental Design
- Vectors
- Kinematics
- Forces
- Current Electricity Fundamentals
- Vectors
Exam Relevance
Measurement concepts are fundamental across H2 Physics. Students are frequently tested on dimensional consistency, experimental uncertainty, systematic and random errors, graph interpretation, significant figures, and vector operations. These skills also support later topics such as vectors, kinematics, and dynamics.
Common exam traps include:
- using the gram instead of kilogram as the SI base unit of mass;
- treating a dimensionally valid equation as automatically correct;
- confusing systematic error with random error;
- claiming too many significant figures from a calculator;
- adding percentage uncertainties for addition or subtraction;
- adding absolute uncertainties for multiplication or division;
- ignoring direction when working with vector quantities.
Links
- Related: vectors
- Related: kinematics
- Related: Thermal Physics A
- Related: current electricity fundamentals
- Related: measurement units and dimensions
- Related: measurement data presentation
- Related: measurement uncertainty and errors
- Related: uncertainty propagation methods
- Related: measurement estimation and experimental design
- Related: scalars vectors and vector operations
Provenance
- source file: 1_PDFsam_01_Measurement.pdf
- generated by:
bridging_tools/ingest_JC_phy_wiki.py - manifest entry:
inbox/lecture_notes/1_PDFsam_01_Measurement.pdf - source hash:
f6acbd161d261405eabedc8d61ebe91cc9b2e187cc1345bbe06c7233d22d2be3