Thermal Measurement and Scales
Overview
This page focuses on how temperature is defined and measured. Since temperature cannot be measured directly, we use physical properties that vary with temperature and calibrate them against agreed reference points.
Core ideas:
- temperature and thermal equilibrium
- thermometric properties
- thermometer calibration
- Celsius scale
- constant-volume gas thermometer
- absolute zero
- Kelvin scale
- zeroth law of thermodynamics
This topic supports the wider chapter:
Definition
Temperature measurement is the process of using a measurable thermometric property and a calibrated scale to assign a numerical value to the thermal state of a body.
Why It Matters
This topic matters because thermal calculations depend on clear distinction between temperature, heat, equilibrium, and absolute temperature. It also explains why kelvin is the preferred physics scale and why thermometers can work at all.
Key Representations
Temperature and Thermal Equilibrium
What is Temperature?
Temperature is a measure of the degree of hotness of a body.
It determines the direction of thermal energy transfer.
If two bodies are placed in contact:
- heat flows from higher temperature to lower temperature
- transfer continues until both reach the same temperature
Thermal Equilibrium
Two objects are in thermal equilibrium when:
- they have the same temperature
- there is no net heat transfer between them
This idea allows thermometers to work.
Heat vs Temperature
Students often confuse these ideas.
Temperature
- indicates hotness
- measured in (^{\circ}\mathrm{C}) or (\mathrm{K})
Heat
- energy transferred due to temperature difference
- measured in joules
A hot object does not necessarily contain more heat than a larger cooler object.
Thermometric Properties
A thermometric property is a measurable physical property that changes with temperature.
Examples:
- length of metal strip
- volume of liquid
- pressure of gas
- electrical resistance
- e.m.f. of thermocouple
Desirable Thermometric Properties
A good thermometric property should be:
- easy to measure
- sensitive to temperature changes
- reproducible
- approximately linear with temperature
- stable over operating range
- unaffected by unrelated factors
Common Thermometers
Liquid-in-Glass Thermometer
Uses expansion of liquid such as mercury or alcohol.
Advantages:
- simple
- inexpensive
Limitations:
- slow response
- limited range
- may be non-linear
Resistance Thermometer / Thermistor
Uses resistance change with temperature.
Advantages:
- sensitive
- suitable for electronic systems
Gas Thermometer
Uses gas pressure or volume.
More fundamental and useful in defining absolute temperature.
Fixed Points and Calibration
To create a scale, two reference temperatures are chosen.
Traditional Celsius scale uses:
- ice point = (0^\circ\mathrm{C})
- steam point = (100^\circ\mathrm{C})
These correspond to melting ice and boiling water under standard atmospheric pressure.
Celsius Calibration
Suppose a thermometer property (x) has:
- (x_0) at ice point
- (x_{100}) at steam point
- (x) at unknown temperature
Then:
This assumes linear variation of the property.
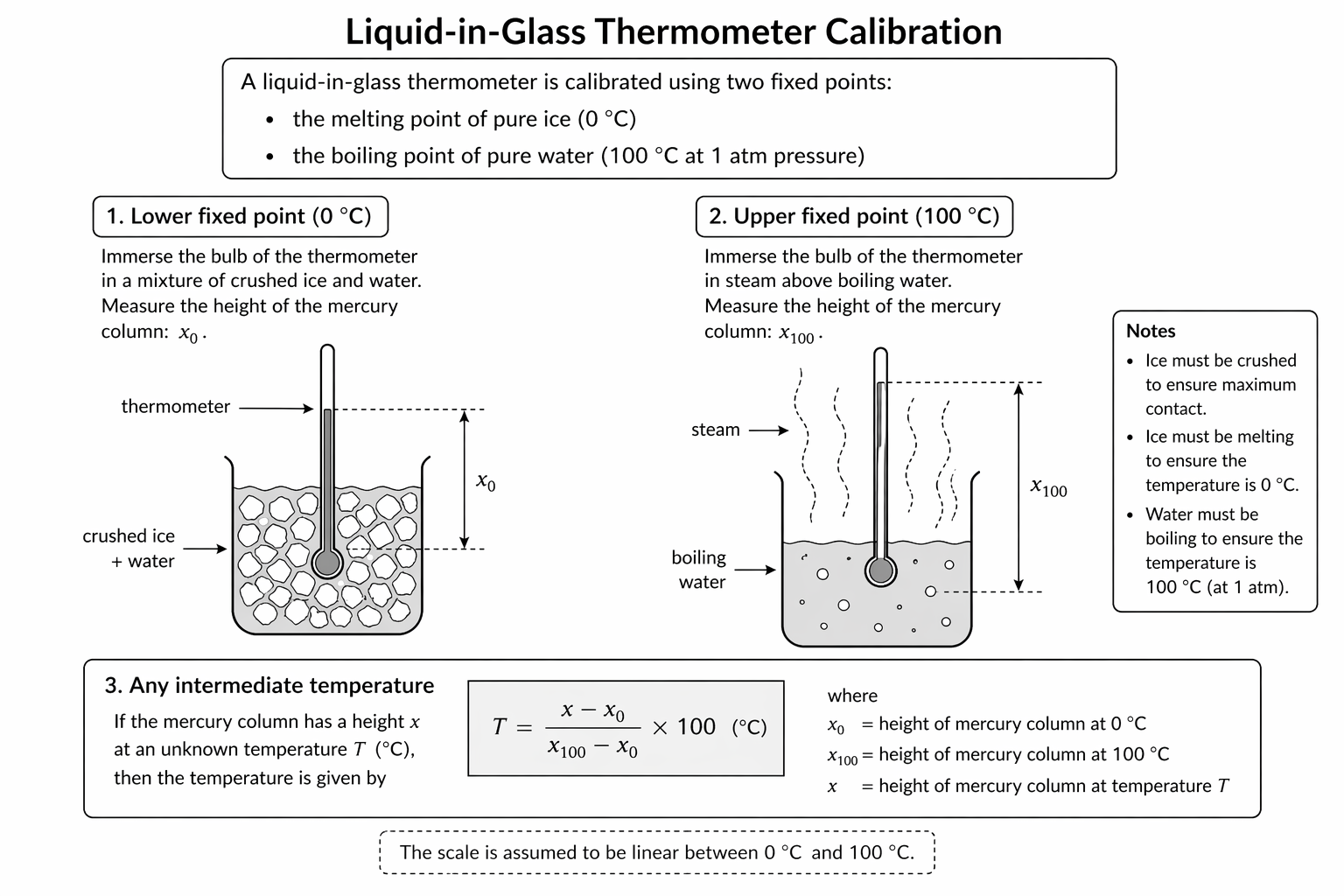
Figure: Calibration of a liquid-in-glass thermometer using lower and upper fixed points, with linear interpolation for an intermediate temperature.
The key assumption is that the thermometric property varies linearly between the two fixed points.
Worked Example 1
A mercury column has:
- (x_0=2.0\ \mathrm{cm})
- (x_{100}=24.0\ \mathrm{cm})
Unknown reading:
- (x=18.0\ \mathrm{cm})
Find temperature.
General Calibration Formula
If lower and upper fixed points are not 0 and 100:
Useful for resistance thermometers or custom scales.
Worked Example 2
A resistance thermometer gives:
- (20\ \Omega) at (-10^\circ\mathrm{C})
- (500\ \Omega) at (110^\circ\mathrm{C})
Find temperature when resistance is (360\ \Omega).
Constant-Volume Gas Thermometer
Principle
For a fixed mass of gas at constant volume:
- pressure increases with temperature
So gas pressure can be used as a thermometric property.
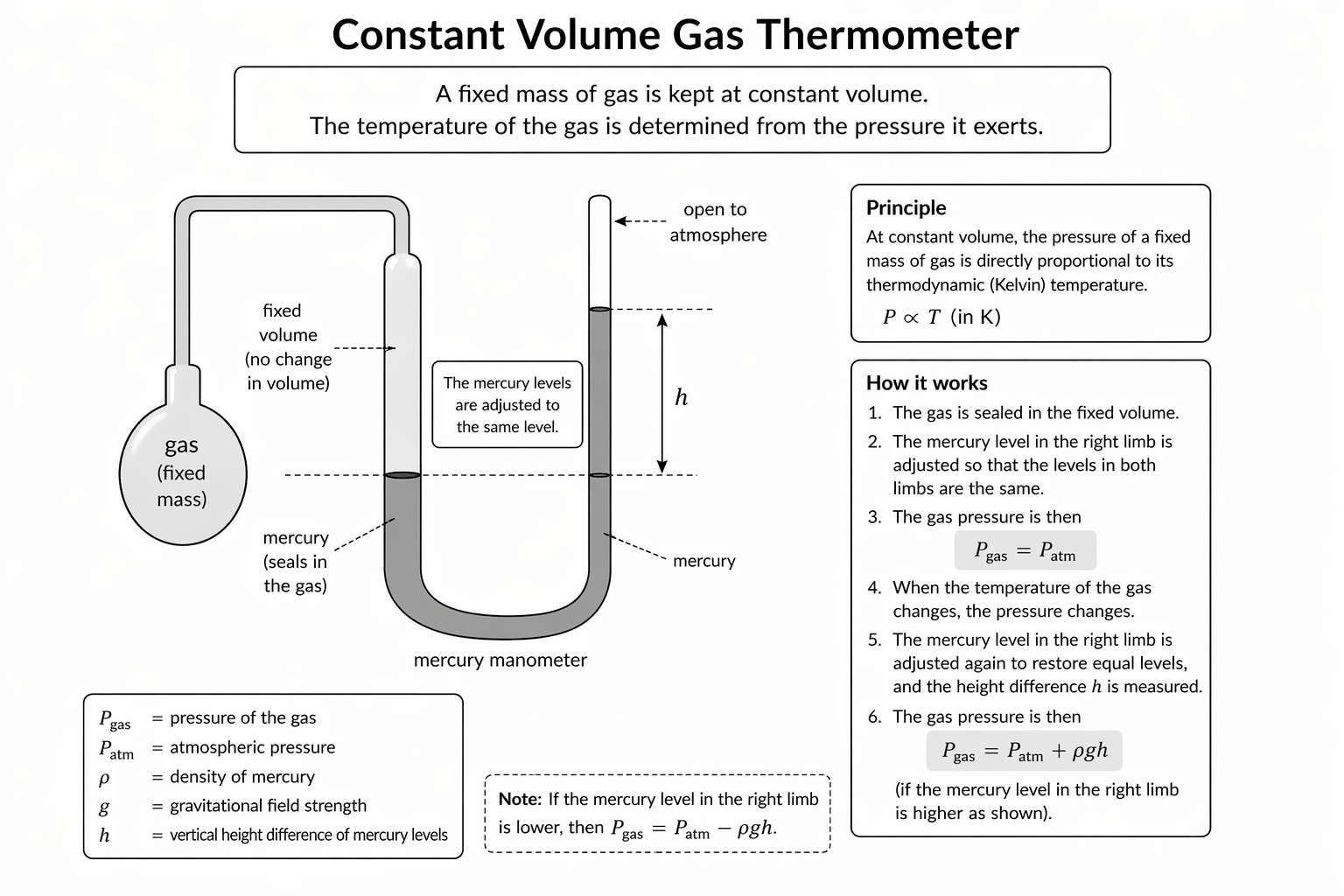
Figure: A constant-volume gas thermometer uses the pressure of a fixed mass of gas as the thermometric property while the gas volume is kept constant.
In practice, the mercury levels are adjusted to keep the gas volume fixed, and the resulting pressure is inferred from the height difference.
Why Important?
Gas behaviour becomes nearly universal at low pressure, making it more reliable than material expansion thermometers.
Pressure-Temperature Graph
At constant volume:
When pressure is plotted against Celsius temperature, extrapolation gives zero pressure near:
This leads to the idea of absolute zero.
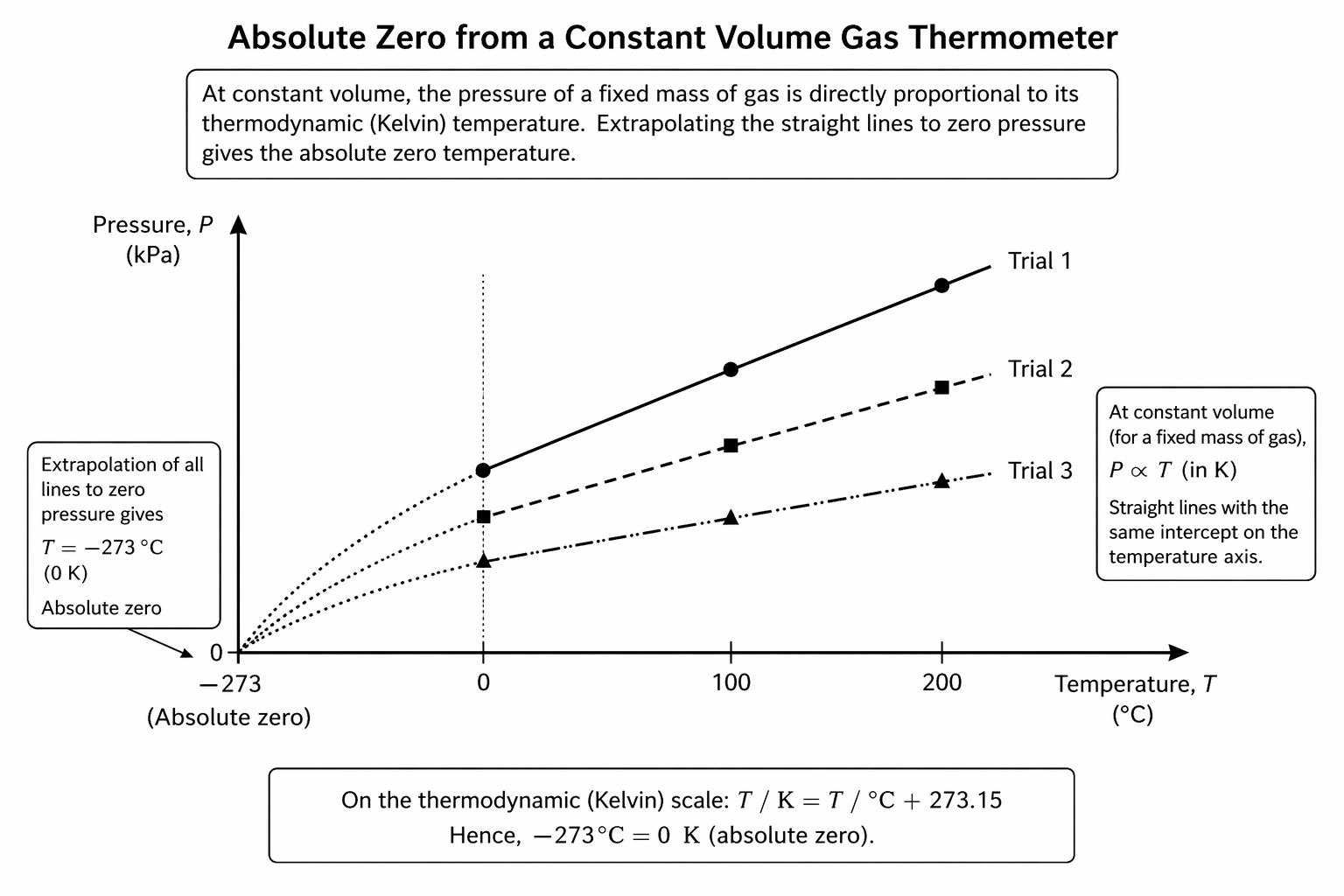
Figure: Extrapolating pressure-temperature lines for a constant-volume gas thermometer leads to the common intercept at absolute zero.
The important exam idea is that different gas samples give different lines, but the extrapolated zero-pressure intercept is the same.
Absolute Zero
Absolute zero is:
- the lowest possible thermodynamic temperature
- temperature where substances have minimum internal energy
Value:
Kelvin Scale
The Kelvin scale is an absolute scale based on thermodynamic principles.
Conversion:
Examples:
- (25^\circ\mathrm{C}=298.15\ \mathrm{K})
- (0^\circ\mathrm{C}=273.15\ \mathrm{K})
Notes on Kelvin
- no degree sign
- cannot be negative in ordinary thermodynamics
- same interval size as Celsius
So:
- rise of (5^\circ\mathrm{C}) = rise of (5\mathrm{K})
Zeroth Law of Thermodynamics
If:
- A is in thermal equilibrium with C
- B is in thermal equilibrium with C
Then:
- A is in thermal equilibrium with B
This allows a thermometer (object C) to compare temperatures of different bodies.
Why Celsius is Less Fundamental
Celsius scale depends on properties of water:
- melting point
- boiling point
These can vary slightly with pressure and impurities.
Kelvin scale is preferred in physics because it is absolute.
Worked Example 3
Convert:
(a) (37^\circ\mathrm{C})
(b) (400\ \mathrm{K})
Experimental Notes
Ice Point
Use:
- pure melting ice
- mixture of ice and water
Steam Point
Use:
- pure steam above boiling water
- standard atmospheric pressure
If conditions are poor, calibration errors occur.
Links
- Thermal Physics A
- Heat Capacity and Latent Heat
- Thermal Practicals
- Thermal Physics A Common Exam Traps
Summary
You should be able to explain:
- what temperature means
- thermal equilibrium
- thermometric properties
- thermometer calibration
- constant-volume gas thermometer
- absolute zero
- Kelvin conversion
- zeroth law of thermodynamics