First Law and Thermodynamic Processes
Overview
This page focuses on how gases exchange energy through heating and mechanical work. It develops the first law of thermodynamics and applies it to standard gas processes commonly tested at Singapore H2 Physics level.
You should be able to:
- distinguish heat, work and internal energy
- use correct sign conventions
- solve first-law questions
- analyse isochoric, isobaric, isothermal and adiabatic changes
- connect equations to particle behaviour
Related hub:
Definition
The first law of thermodynamics is the energy-conservation statement for thermal systems: internal energy changes because of heat transfer and work.
Why It Matters
This topic is where many sign errors happen. Students often know the formula but lose marks by not stating whether work is done on the gas or by the gas, or by confusing isothermal with adiabatic changes.
Key Representations
1. Internal Energy Revisited
Internal energy (U) is the total microscopic energy stored in a system.
It consists of:
- random kinetic energy of particles
- intermolecular potential energy
For an ideal gas:
- intermolecular forces are negligible
- potential energy is approximately zero
Hence:
For a monatomic ideal gas:
So internal energy depends only on temperature.
2. Heat, Work and Internal Energy
Heat (Q)
Heat is energy transferred because of a temperature difference.
- energy into system: (Q>0)
- energy out of system: (Q<0)
Work (W)
In this chapter, (W) means work done on the gas.
- compression by surroundings: (W>0)
- expansion by gas: (W<0)
Internal Energy Change
- increase in internal energy: (\Delta U>0)
- decrease in internal energy: (\Delta U<0)
3. First Law of Thermodynamics
Statement
The increase in internal energy of a system equals heat supplied to the system plus work done on the system.
This is an application of conservation of energy.
Rearranged Forms
Use whichever form is most convenient.
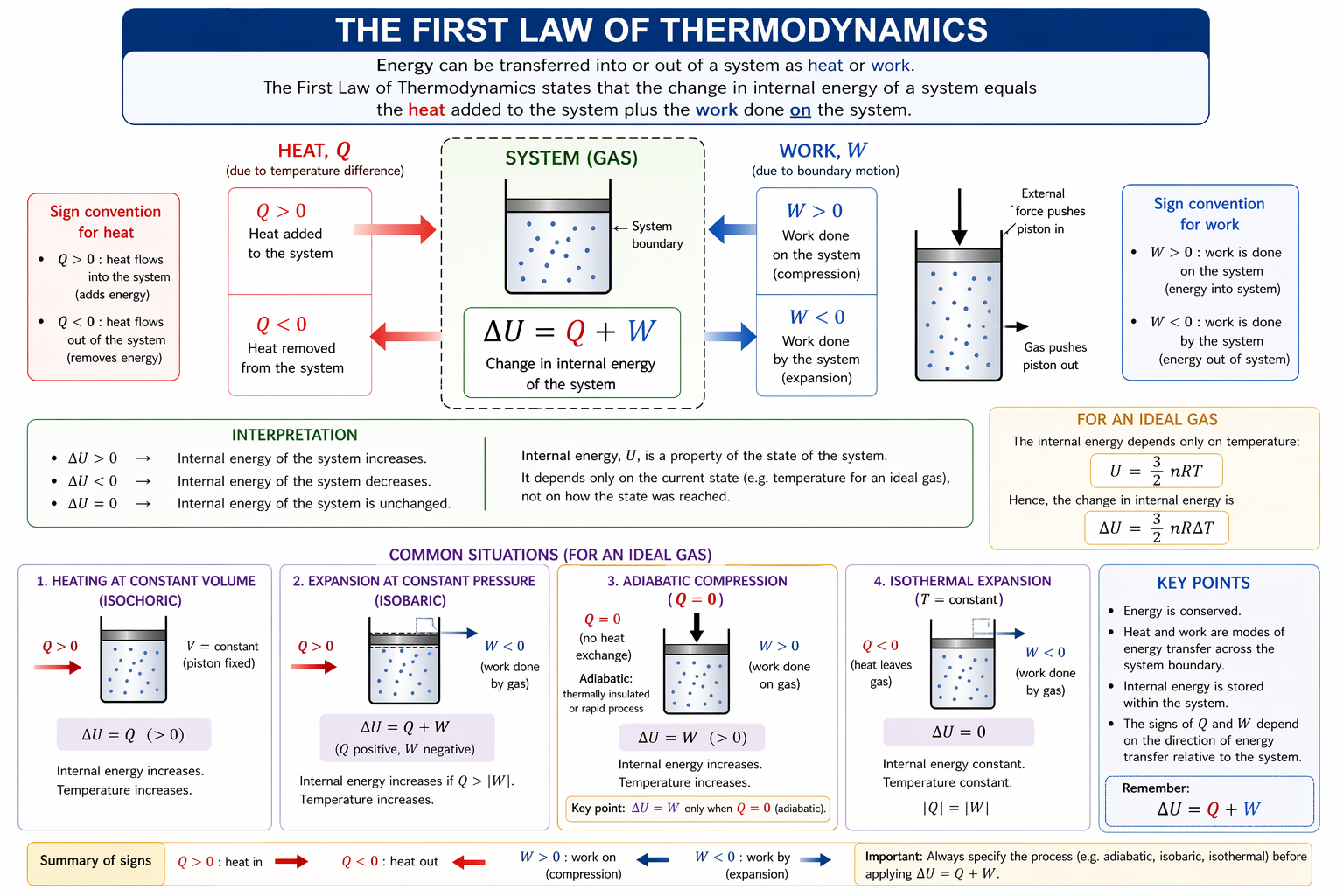
Figure: The first law links internal energy change to heat transfer and work, using the sign convention ( \Delta U = Q + W ) with work done on the gas taken as positive.
Use this sign convention consistently before substituting numbers; most first-law mistakes come from mixing “work done on gas” and “work done by gas”.
4. Work Done by Gas
General Formula
Work done on gas:
Meaning of Sign
Compression
Volume decreases:
So:
Surroundings transfer energy mechanically into gas.
Expansion
Volume increases:
So:
Gas transfers energy to surroundings.
5. Constant Pressure Work
If pressure is constant:
Where:
Cases
Expansion
Compression
6. Standard Thermodynamic Processes
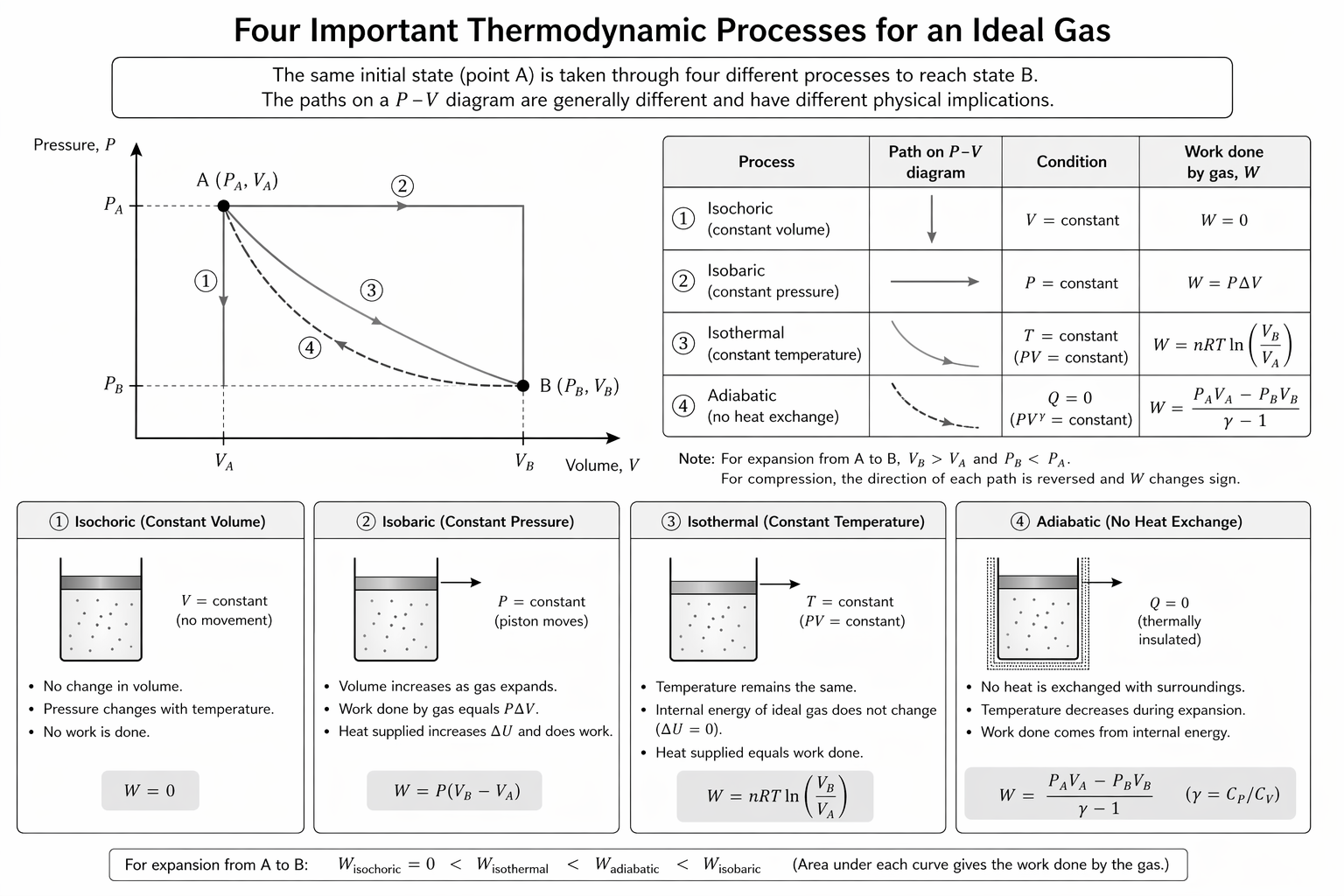
Figure: The four standard ideal-gas processes compared on a p-V diagram: isochoric, isobaric, isothermal, and adiabatic.
This comparison is most useful as a quick process-identification map: first state what stays constant, then simplify the first law accordingly.
6.1 Isochoric Process (Constant Volume)
Condition
Therefore:
No boundary movement, so:
Hence first law becomes:
Interpretation
All heat supplied changes internal energy.
For Ideal Gas
- temperature rises if heat is supplied
- pressure rises because molecules move faster
6.2 Isobaric Process (Constant Pressure)
Condition
Work done:
First law:
Interpretation
Heat supplied may be used for:
- increasing internal energy
- doing expansion work
6.3 Isothermal Process (Constant Temperature)
Condition
For ideal gas:
Hence:
Therefore:
Interpretation
Any heat supplied is converted into work done by gas.
Any work done on gas leaves as heat.
Shape on p-V Graph
Rectangular hyperbola:
6.4 Adiabatic Process
Condition
No heat exchange:
Hence:
Interpretation
Compression
- (W>0)
- internal energy rises
- temperature rises
Expansion
- (W<0)
- internal energy falls
- temperature falls
How Achieved
- thermal insulation
- very rapid process (little time for heat transfer)
7. Process Summary Table
| Process | Constant Quantity | Work Done on Gas | Internal Energy |
|---|---|---|---|
| Isochoric | (V) | (0) | (\Delta U=Q) |
| Isobaric | (P) | (-P\Delta V) | depends |
| Isothermal (ideal gas) | (T) | non-zero | (\Delta U=0) |
| Adiabatic | (Q=0) | non-zero | (\Delta U=W) |
8. Particle Explanations
Heating at Constant Volume
- molecules move faster
- more energetic wall collisions
- pressure increases
Compression
- walls push molecules inward
- work transferred into gas
- temperature often rises
Expansion
- gas pushes piston outward
- gas loses energy as work
- temperature may fall if no heat enters
9. Worked Examples
Example 1: Heat Supplied + Compression
A gas receives (120,\text{J}) heat.
Work done on gas is (40,\text{J}).
Find (\Delta U).
Internal energy increases by (160,\text{J}).
Example 2: Gas Expands Doing Work
A gas absorbs (200,\text{J}) heat and does (70,\text{J}) work.
Since gas does work:
Then:
Example 3: Constant Volume Heating
Gas at constant volume absorbs (90,\text{J}).
Since:
Then:
Example 4: Isothermal Expansion
Ideal gas expands isothermally and does (50,\text{J}) work.
So:
Since:
Then:
Heat absorbed is (50,\text{J}).
Example 5: Adiabatic Compression
Gas compressed adiabatically with work done on gas (75,\text{J}).
Since:
Temperature increases.
10. Strategy for First-Law Questions
Step 1: Identify Sign Convention
Use:
- heat into gas positive
- work on gas positive
Step 2: Determine Process Type
Look for:
- constant volume
- constant pressure
- constant temperature
- insulated or adiabatic
Step 3: Apply Simplification
Examples:
- constant volume → (W=0)
- isothermal ideal gas → (\Delta U=0)
- adiabatic → (Q=0)
Step 4: Substitute Carefully
Links
- Thermal Physics B
- Kinetic Theory and Ideal Gases
- p-V Diagrams and Cycles
- Thermal Physics B Common Exam Traps
- Work, Energy and Power
Summary
The first law becomes much easier once the process type is clear. The real skill is identifying what is zero, what changes sign, and which quantity is a state property.